Checkmate 81612/30/2023 This decision marks the fourth tumor type in which Opdivo -based regimens have been approved for use in earlier stages of cancer in the European Union, following melanoma, esophageal/gastroesophageal junction cancer and urothelial carcinoma Opdivo with chemotherapy is now the first and only neoadjuvant immunotherapy-based treatment option approved to treat patients with non-small cell lung cancer in the European Union 
Philadelphia (PA): AACR Cancer Res 2021 81(13_Suppl):Abstract nr CT003.Approval based on results from Phase 3 CheckMate -816 trial showing that neoadjuvant Opdivo with chemotherapy improved event-free survival and pathologic complete response compared to chemotherapy alone In: Proceedings of the American Association for Cancer Research Annual Meeting 2021 2021 Apr 10-15 and May 17-21. Nivolumab (NIVO) + platinum-doublet chemotherapy (chemo) vs chemo as neoadjuvant treatment (tx) for resectable (IB-IIIA) non-small cell lung cancer (NSCLC) in the phase 3 CheckMate 816 trial. Saylors, Fumihiro Tanaka, Hiroyuki Ito, Ke-Neng Chen, Cecile Dorange, Junliang Cai, Joseph Fiore, Nicholas Girard. Swanson, Keith Kerr, Changli Wang, Gene B. Awad, Enriqueta Felip, Stephen Broderick, Julie Brahmer, Scott J. Forde, Jonathan Spicer, Shun Lu, Mariano Provencio, Tetsuya Mitsudomi, Mark M. 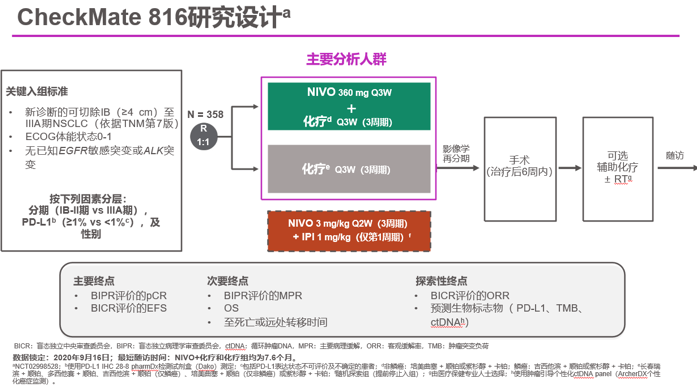
CheckMate 816 is the first positive phase 3 trial demonstrating a significant improvement in pathologic response with neoadjuvant immunotherapy plus chemo in resectable NSCLC.Ĭitation Format: Patrick M. The safety profile of NIVO + chemo was consistent with the known profile of this combination regimen, and the addition of NIVO did not decrease the ability to perform surgery. Grade 3-4 tx-related AEs and grade 3-4 surgery-related AEs were reported in 33.5% vs 36.9% and 11.4% vs 14.8% of pts in the NIVO + chemo vs chemo arms, respectively.Ĭonclusions CheckMate 816 met its first primary endpoint with a statistically significant improvement in pCR with neoadjuvant NIVO + chemo vs chemo alone per independent review. Definitive surgery occurred for 83.2% of pts treated with NIVO + chemo and 75.4% with chemo surgery was cancelled rarely due to AEs (2 pts/arm) and due to disease progression in 12 and 17 pts, respectively. NIVO + chemo also improved MPR rates vs chemo in the ITT (36.9% vs 8.9%), as well as ORR (53.6% vs 37.4%) and radiographic down-staging rates (30.7% vs 23.5%). Improvement in pCR with NIVO + chemo vs chemo was consistent across key subgroups including disease stage (IB/II ≥ IIIA ), PD-L1 (< 1% ≥ 1% ), and TMB (low high ). Neoadjuvant NIVO + chemo significantly increased pCR rates vs chemo in the intent-to-treat population (ITT) (24.0% vs 2.2% odds ratio 13.94 P < 0.0001). Results Baseline characteristics were balanced between arms (n = 179 each). Key exploratory endpoints are objective response rate (ORR) per BICR and potential predictive biomarkers including PD-L1 and tumor mutational burden (TMB). Overall survival, major pathological response (MPR ≤ 10% viable tumor in both lung and lymph nodes) per BIPR, and time to death or distant metastases are secondary endpoints. pCR was defined as 0% viable tumor cells in resected lung and lymph nodes pts who did not undergo surgery were counted as non-responders. pCR by blinded independent pathological review (BIPR) and event-free survival by blinded independent central review (BICR) are the primary endpoints. Stratification was by disease stage (IB/II vs IIIA), PD-L1 (≥ 1% or < 1%), and sex. 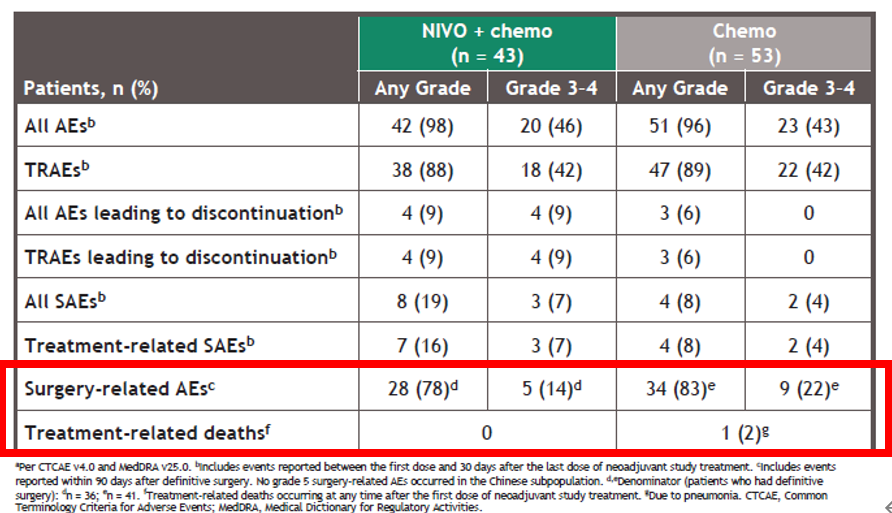
Methods Adults with clinical stage IB (≥ 4 cm)-IIIA (per AJCC 7 th ed), resectable NSCLC, ECOG PS 0-1, and no known EGFR/ALK alterations were randomized to either NIVO 360 mg + platinum-doublet chemo Q3W or chemo Q3W for 3 cycles, followed by surgery. Here, we report the final analysis of one of the primary endpoints, pCR, of CheckMate 816 (NCT02998528)-a randomized, phase 3, open-label study evaluating NIVO + chemo vs chemo as neoadjuvant tx for resectable NSCLC. 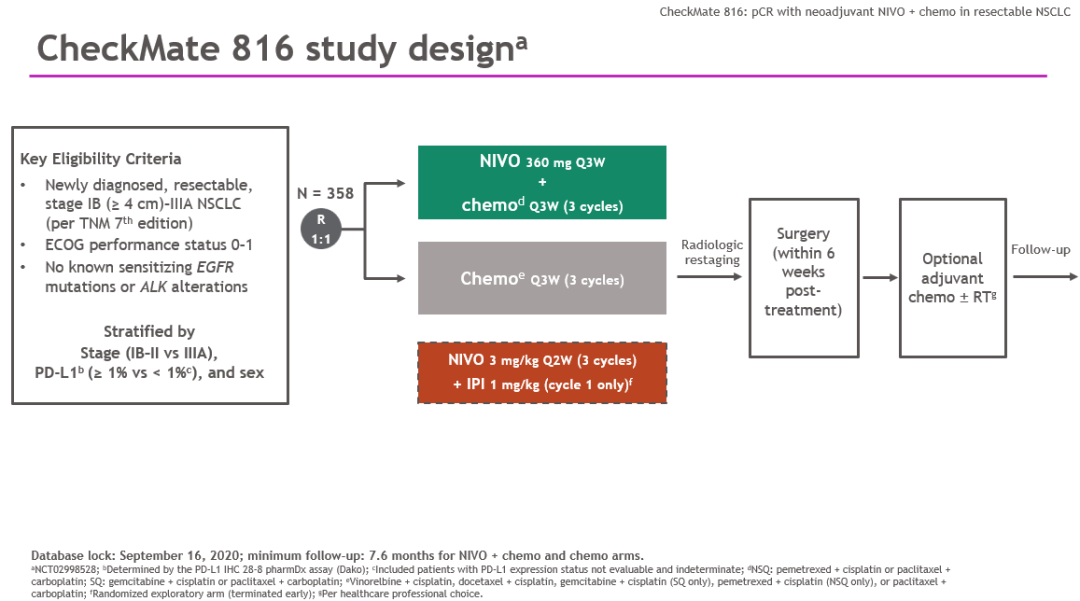
Recently, neoadjuvant NIVO, alone or in combination with chemo, has shown encouraging pCR rates in single-arm phase 2 studies. Although immunotherapy targeting the PD-1 pathway has shown survival benefits in metastatic NSCLC, phase 3 trial results in resectable disease are yet to be reported. Neoadjuvant or adjuvant chemo is recommended for pts with high recurrence risk however, benefits are modest and pathological complete response (pCR) with neoadjuvant chemo is low. Background In patients (pts) with non-metastatic NSCLC, surgery has curative potential but 30-80% who undergo resection experience recurrence.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
